cartriQ® BioPure Ready-to-use pharma glass cartridges with superior drug stability
- Type I Borosilicate Glass cartridge for unmatched patient safety and safe self-administration
- Reduced risk of pH shift and high formulation integrity for sensitive drugs
- No heavy metals according to ICH Q3D Class 1, 2, and 3
- Tight dimensional tolerances and cosmetic defect limits for reliable functionality and integrity
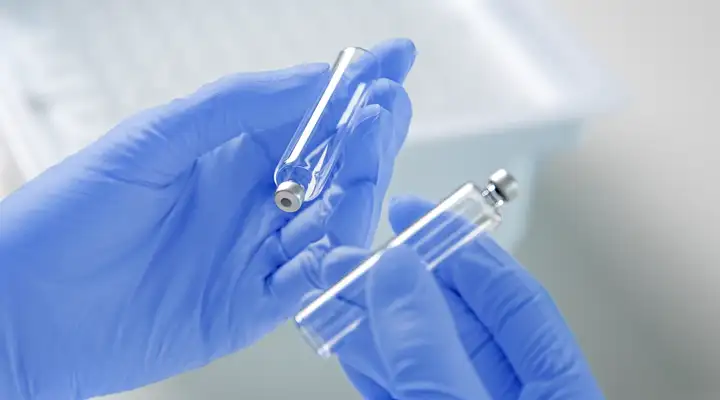
cartriQ® BioPure: Your first choice for pharmaceutical ready-to-use glass cartridges
Biotech pipelines are increasingly defined by complex molecules, peptides, and next‑generation modalities that demand reliable, low‑risk primary packaging. Conventional ready‑to‑use glass cartridges can fall short, leading to elevated leachable levels, pH shifts, and increased aggregation risk.
SCHOTT Pharma’s cartriQ® BioPure is designed to address these challenges. Combining FIOLAX® Pro Type I Borosilicate Glass with the latest cartridge manufacturing technology and steam‑sterilization process, this glass cartridge offers a highly uniform inner surface with enhanced hydrolytic resistance and significantly reduced leachable values. For the first time, this superior packaging solution is paired with strict release criteria for sodium and aluminum content.
Part of the SCHOTT iQ® platform, cartriQ® BioPure cartridges are delivered in an industry‑standard nest‑and‑tub configuration, simplifying fill‑and‑finish operations and reducing process complexity. Combined with high‑performance coated and uncoated elastomers from SCHOTT Pharma’s broad partner network, the system allows innovators to focus on the molecule rather than the container and to evaluate multiple device and elastomer options without changing the core container.
Reliable choice from the start
Made from FIOLAX® Pro Type I Borosilicate Glass, with its exceptionally high hydrolytic resistance and aluminum levels matching the current USP<660> draft guidance, cartriQ® BioPure is the gold standard for patient safety. It provides long‑term stability for highly sensitive biologics, enabling biotech firms to focus on future formulation adjustments and improved stability performance as complexity increases.
Every cartriQ® BioPure glass cartridge undergoes rigorous leachables testing throughout its production, helping to maintain the integrity of sensitive APIs, regardless of the fill volume. Its tight dimensional tolerances and controlled manufacturing process are engineered to interact, ensuring container closure integrity and high dosing accuracy from early development to commercial scale.
Quality‑by‑design glass cartridges for sensitive biologics
cartriQ® BioPure glass cartridges are engineered to meet the strict requirements of high‑value biologics. Designed according to the principles of quality‑by‑design, from raw materials to manufacturing, the optimized inner surface enhances formulation stability while minimizing the risk of time‑consuming reformulations, including those caused by known critical buffers.
The glass cartridges are ideal for the storage and self‑administration of emerging biologics and biosimilars used in handheld injection devices, such as peptides and recombinant proteins, rare and orphan drugs, emergency medicines, and high‑value formulations with limited API availability.
cartriQ® BioPure cartridges are available pre‑sterilized and delivered ready‑to‑use in an industry‑standard nest‑and‑tub configuration, enabling highly efficient fill‑and‑finish processes.


|

|

|

|

|

|
|
| Product Name |
Product Name
cartriQ® BioPure
|
Product Name
cartriQ®
|
Product Name
cartriQ® Large Volume
|
Product Name
Core Cartridges
|
Product Name
Cartridges Break Resistant
|
Product Name
Cartridges Double Chamber
|
| Applications |
Applications
Washed and pre-sterilized for highly sensitive biologics
|
Applications
Pre-washed and pre-sterilized for small-volume biologics
|
Applications
Pre-washed and pre-sterilized for large-volume biologics
|
Applications
Default solution for generics, chemicals and biologics
|
Applications
For emergency drugs
|
Applications
For lyophilized drugs
|
| Tubing Container Material |
Tubing Container Material
FIOLAX® Pro
|
Tubing Container Material
FIOLAX® CHR (controlled hydrolytic resistance)
|
Tubing Container Material
FIOLAX® CHR (controlled hydrolytic resistance)
|
Tubing Container Material
FIOLAX®
|
Tubing Container Material
FIOLAX®
|
Tubing Container Material
FIOLAX®
|
| Format |
Format
3 ml, 5 ml
|
Format
1.5 - 3 ml
|
Format
5 ml, 10 ml
|
Format
1.5 - 20 ml
|
Format
Customized upon request
|
Format
Customized upon request
|
| Quality Level |
Quality Level
TopLine
|
Quality Level
TopLine
|
Quality Level
TopLine
|
Quality Level
StandardLine, TopLine
|
Quality Level
StandardLine
|
Quality Level
StandardLine
|
| Closure Systems |
Closure Systems
Stopper + Seal
|
Closure Systems
Stopper + Seal
|
Closure Systems
Stopper + Seal
|
Closure Systems
Stopper + Seal
|
Closure Systems
Stopper + Seal
|
Closure Systems
Stopper + Seal
|
| Pre-washed, Pre-sterilized |
Pre-washed, Pre-sterilized
Sterile available
|
Pre-washed, Pre-sterilized
Sterile available
|
Pre-washed, Pre-sterilized
Sterile available
|
Pre-washed, Pre-sterilized
Non-sterile and sterile (cartriQ®) available
|
Pre-washed, Pre-sterilized
Non-sterile available
|
Pre-washed, Pre-sterilized
Non-sterile available
|
| Packaging |
Packaging
Pre-sterilized and pre-washed cartridges in cup nest
|
Packaging
Pre-sterilized and pre-washed cartridges in cup nest
|
Packaging
Pre-sterilized and pre-washed cartridges in cup nest
|
Packaging
Bulk tray, cartriQ® solution (cup nest)
|
Packaging
Bulk tray
|
Packaging
Bulk tray
|
| DATASHEET | DATASHEET | STANDARDLINE TOPLINE | ||||
| Read More | Read More | Read More | Read More | Read More |











