First large-volume glass syringes for subcutaneous injections
syriQ BioPure® 5.5 ml glass syringe for large-volume biologics
As biologic therapies advance toward higher volumes and more complex formulations, the demand for delivery systems that support patient-friendly self-administration continues to grow. Larger subcutaneous injections are becoming increasingly prominent, driven by innovations in formulation science and the need to simplify drug administration across new therapeutic areas.Successfully developing large-volume drug delivery systems requires balancing pharmaceutical requirements, user needs, processability, and business goals. Thinking in systems and understanding the critical interfaces of glass prefilled syringes are essential to ensure safe, stable, and reliable delivery, especially for high-viscosity biologics.
Are you ready for large-volume biologics in ready-to-use glass syringes?
SCHOTT Pharma’s syriQ BioPure® 5.5 ml large-volume staked-needle glass syringe addresses the growing demand for subcutaneous delivery of large-volume biologics in a single dose, reducing the need for multiple injections while improving patient comfort. Engineered with ultra-low tungsten and glue residuals, tight dimensional tolerances, and a homogeneous silicone layer, the glass syringe ensures long-term drug stability and minimizes interaction risks, even for highly sensitive and viscous formulations.This innovation reflects a platform-based approach that enables scalable homecare solutions and helps pharma partners accelerate market entry while maintaining the highest standards of safety and performance.

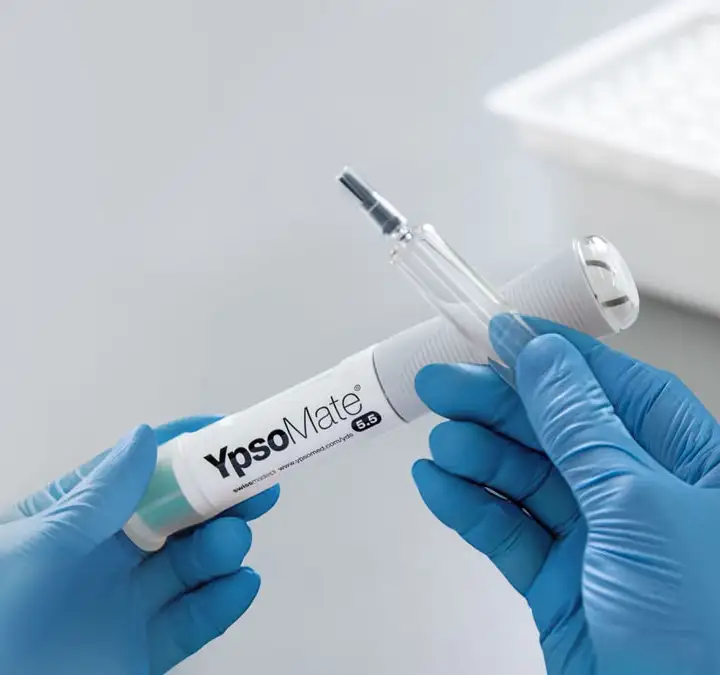
Seamless integration of syriQ BioPure® 5.5 ml for reliable subcutaneous delivery
The new large-volume staked-needle glass syringe is available in 5.5 ml format for autoinjector use, and has been co-developed with the large-volume autoinjector YpsoMate® 5.5 from Ypsomed.Partnering for success with SCHOTT Pharma
SCHOTT Pharma offers a comprehensive analytical testing suite to streamline packaging-related testing during clinical trials and drug submission. Through PartnerLab, we provide a broad range of testing services tailored to meet the diverse and stringent requirements of drug product packaging and interaction testing. PartnerLab helps streamline your testing process, accelerate time-to-market, and elevates the assurance of your product’s quality and safety. We also support troubleshooting during critical development stages.
In addition, our fill-and-finish services enables smooth early-stage sample filling for initial testing. To support long-term success, we offer filling line compatibility verification, including assessments of your specific line and efficiency optimization. Moreover, we ensure the global regulatory availability of our containment solutions in compliance with evolving international norms and standards, as well as country-specific laws and guidelines for a fast time-to-market.



