
Core Vials Pharmaceutical glass vials in StandardLine and TopLine quality
BUY ONLINE NOWSetting the industry standard
The pharmaceutical manufacturing industry is under increasing pressure to navigate rising costs, strict regulations, and complex operations. To address these challenges head-on, SCHOTT Pharma delivers a targeted, high-quality solution: Core Vials.
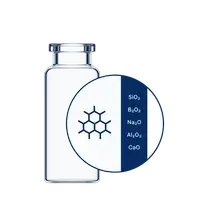
As a trusted choice for injectable drugs, vials are essential for ensuring safe, long-term storage, with just the glass and stoppers coming into direct contact with the medication. Core Vials set a new standard with their uniform dimensional accuracy and high cosmetic quality. These advanced properties empower you to achieve higher yields, lower Total Cost of Ownership (TCO), and minimize waste of precious drug products. Core Vials are available in three quality levels: StandardLine, TopLine and TopLine Packages.
- StandardLine: Minimum cosmetic accepted quality levels (AQLs) according to the current Defect Evaluation List (DEL) and defined as the most relevant industry standard (also aligned with current PDA Technical Report).
- TopLine: Tighter cosmetic AQLs than StandardLine.
- TopLine Packages: Provide even tighter AQLs for dedicated fill-and-finish challenges.
Choosing inferior, low-cost containers often leads to hidden costs such as production delays, increased rejection rates, and compromised drug integrity. SCHOTT Pharma’s 4,600 specialists work tirelessly to refine production processes and ensure exceptional container uniformity, delivering solutions that consistently meet the highest industry standards.
Optimize savings and minimize Total Cost of Ownership (TCO)
Increase your drug production yield and experience low TCO with smooth line performance with SCHOTT Pharma Core Vials. Their uniform dimensions and high cosmetic quality have a positive impact on your fill-and-finish line efficiency, leading to fewer container-induced downtime and low reject rates of finished drug product.
When to use Core Vials
SCHOTT Pharma Core Vials are a safe and efficient containment solution for storing chemical drugs and generics, including essential medicines, traditional vaccines, and veterinary products.
TopLine Packages, serving as Upgrades for Core Vials in TopLine quality, help streamline your fill-and-finish operations with precision and reliability, and can be freely combined in any configuration to suit your specific needs.
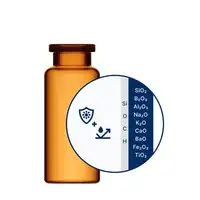
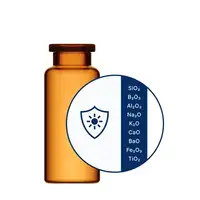
For especially demanding drugs, SCHOTT Pharma’s EVERIC® portfolio provides a superior solution.


|
|

|

|

|

|

|

|
|
|

|
|
| Product Name |
Product Name
Core Vials
|
Product Name
EVERIC® pure
|
Product Name
EVERIC® care
|
Product Name
EVERIC® freeze
|
Product Name
EVERIC® strong & smooth
|
Product Name
EVERIC® smart
|
Product Name
EVERIC® plus
|
Product Name
EVERIC® lyo
|
Product Name
EVERIC® lyo & amber
|
Product Name
EVERIC® amber
|
Product Name
adaptiQ®
|
| Applications |
Applications
Default solution for generics, chemicals
|
Applications
Default solution for biologics, diluents, low-filled and high-value drugs
|
Applications
Drugs in high alkaline pH range
|
Applications
Drugs that need deep-cold storage down to -80°
|
Applications
Improved bulk fill-and-finish
|
Applications
Track and trace solution
|
Applications
Leachable-sensitive drugs in neutral and acidic pH range
|
Applications
Drugs that need lyophilization
|
Applications
Light sensitive, lyophilized drugs, esp. biologics as ADCs
|
Applications
Light-sensitive drugs, esp. biologics as ADCs
|
Applications
Pre-washed and pre-sterilized for leaner fill-and-finish
|
| Tubing Container Material |
Tubing Container Material
FIOLAX®
|
Tubing Container Material
FIOLAX® CHR (controlled hydrolytic resistance)
|
Tubing Container Material
FIOLAX® CHR (controlled hydrolytic resistance)
|
Tubing Container Material
FIOLAX® OS (optimized strength)
|
Tubing Container Material
FIOLAX® OS (optimized strength)
|
Tubing Container Material
FIOLAX®
|
Tubing Container Material
FIOLAX®
|
Tubing Container Material
FIOLAX®
|
Tubing Container Material
FIOLAX® amber
|
Tubing Container Material
FIOLAX® amber
|
Tubing Container Material
Depending on bulk vial used
|
| Format |
Format
2-100R
|
Format
2-50R
|
Format
2R, 6R, 10R
|
Format
2-30R
|
Format
2-30R
|
Format
2-30R
|
Format
2-50R
|
Format
2-50R
|
Format
6R, 10R, 20 ml
|
Format
2R - 10R, 20 ml
|
Format
2-50R
|
| Quality Level |
Quality Level
StandardLine, TopLine, TopLine Packages
|
Quality Level
TopLine
|
Quality Level
TopLine
|
Quality Level
TopLine
|
Quality Level
TopLine
|
Quality Level
TopLine
|
Quality Level
TopLine
|
Quality Level
TopLine
|
Quality Level
StandardLine
|
Quality Level
StandardLine
|
Quality Level
TopLine
|
| Closure Systems |
Closure Systems
Stopper + seal
|
Closure Systems
Stopper + seal
|
Closure Systems
Stopper + seal
|
Closure Systems
Stopper + seal
|
Closure Systems
Stopper + seal
|
Closure Systems
Stopper + seal
|
Closure Systems
Stopper + seal
|
Closure Systems
Stopper + seal
|
Closure Systems
Stopper + seal
|
Closure Systems
Stopper + seal
|
Closure Systems
Stopper + seal or press-fit cap
|
| Pre-washed, Pre-sterilized |
Pre-washed, Pre-sterilized
Non-sterile and sterile (only TopLine in adaptiQ®) available
|
Pre-washed, Pre-sterilized
Non-sterile and sterile (adaptiQ®) available
|
Pre-washed, Pre-sterilized
Non-sterile and sterile (adaptiQ®) available
|
Pre-washed, Pre-sterilized
Non-sterile available
|
Pre-washed, Pre-sterilized
Non-sterile available
|
Pre-washed, Pre-sterilized
Non-sterile and sterile (adaptiQ®) available
|
Pre-washed, Pre-sterilized
Non-sterile and sterile (adaptiQ®) available
|
Pre-washed, Pre-sterilized
Non-sterile and sterile (adaptiQ®) available
|
Pre-washed, Pre-sterilized
Non-sterile and sterile* (adaptiQ®) available
|
Pre-washed, Pre-sterilized
Non-sterile and sterile* (adaptiQ®) available
|
Pre-washed, Pre-sterilized
Sterile packaging
|
| Packaging | Packaging | Packaging | Packaging | Packaging | Packaging | Packaging | Packaging | Packaging | Packaging | Packaging | Packaging |
| Read More | Read More | Read More | Read More | Read More | Read More | Read More | Read More | Read More |
*available from Q2 2026









