EVERIC® plus Pharmaceutical glass vials for highly sensitive drugs
- Inner silicon dioxide barrier coating via PICVD reduces leachables
- Can minimize protein adsorption and pH-shift to stabilize drugs
- Suitable for complex proteins, radiopharmaceuticals, and diluents
- Available pre-washed and pre-sterilized ready-to-use configurations

EVERIC® plus vials are the ideal containers for sensitive drug formulations
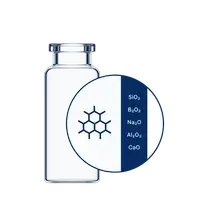
For highly sensitive drugs, leachables from packaging can affect stability, while protein adsorption can cause denaturation, aggregation, and loss of effectiveness. As a solution for highly sensitive drugs, EVERIC® plus vials feature a silicon dioxide barrier coating created using plasma impulse chemical vapor deposition (PICVD) technology that results in covalent bonding.
This silicon dioxide coating minimizes glass leachables in the neutral and acidic pH range, minimizing pH-shift. This can be particularly beneficial when reconstituting drugs using diluents such as water for injection (WFI). In addition, protein adsorption is reduced, enhancing drug stability and effectiveness.
Based on Type I Borosilicate Glass and available in TopLine quality, the additional ion barrier ensures your sensitive drug formulations are safely stored in low-to-medium pH ranges.
Superior drug stability and integrity
The silicon dioxide inner coating of EVERIC® plus vials acts as an ion barrier, providing stability for sensitive drugs in the acidic and neutral pH range. The resulting avoidance of pH-shift also helps to keep diluents such as WFI stable, while minimizing protein adsorption.
Reliable storage solution for complex proteins, radiopharmaceuticals, and diluents
EVERIC® plus vials are suitable for a wide range of applications. Thanks to their low level of leachables, they are ideal for storing complex proteins, radiopharmaceuticals, and diluents.
Reduced levels of leached aluminum also enable the storage of drugs that contain amino acids, such as parenteral nutrition.
Available in pre-washed, pre-sterilized ready-to-use (RTU) configurations, EVERIC® plus vials are compatible with a wide range of fill-and-finish lines and can remain nested throughout the process (including freeze-drying).


|
|

|

|

|

|

|
|
|

|

|
|
| Product Name |
Product Name
EVERIC® plus
|
Product Name
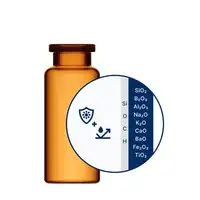
EVERIC® pure
|
Product Name
EVERIC® care
|
Product Name
EVERIC® freeze
|
Product Name
EVERIC® strong & smooth
|
Product Name
EVERIC® smart
|
Product Name
EVERIC® lyo
|
Product Name
EVERIC® lyo & amber
|
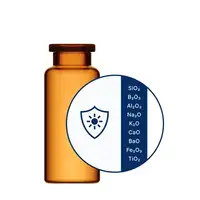
Product Name
EVERIC® amber
|
Product Name
adaptiQ®
|
Product Name
Core Vials
|
| Applications |
Applications
Leachable-sensitive drugs in neutral and acidic pH range
|
Applications
Default solution for biologics, diluents, low-filled and high-value drugs
|
Applications
Drugs in high alkaline pH range
|
Applications
Drugs that need deep-cold storage down to -80°
|
Applications
Improved bulk fill-and-finish
|
Applications
Track and trace solution
|
Applications
Drugs that need lyophilization
|
Applications
Light sensitive, lyophilized drugs, esp. biologics as ADCs
|
Applications
Light-sensitive drugs, esp. biologics as ADCs
|
Applications
Pre-washed and pre-sterilized for leaner fill-and-finish
|
Applications
Default solution for generics, chemicals
|
| Tubing Container Material |
Tubing Container Material
FIOLAX®
|
Tubing Container Material
FIOLAX® CHR (controlled hydrolytic resistance)
|
Tubing Container Material
FIOLAX® CHR (controlled hydrolytic resistance)
|
Tubing Container Material
FIOLAX® OS (optimized strength)
|
Tubing Container Material
FIOLAX® OS (optimized strength)
|
Tubing Container Material
FIOLAX®
|
Tubing Container Material
FIOLAX®
|
Tubing Container Material
FIOLAX® amber
|
Tubing Container Material
FIOLAX® amber
|
Tubing Container Material
Depending on bulk vial used
|
Tubing Container Material
FIOLAX®
|
| Format |
Format
2-50R
|
Format
2-50R
|
Format
2R, 6R, 10R
|
Format
2-30R
|
Format
2-30R
|
Format
2-30R
|
Format
2-50R
|
Format
6R, 10R, 20 ml
|
Format
2R - 10R, 20 ml
|
Format
2-50R
|
Format
2-100R
|
| Quality Level |
Quality Level
TopLine
|
Quality Level
TopLine
|
Quality Level
TopLine
|
Quality Level
TopLine
|
Quality Level
TopLine
|
Quality Level
TopLine
|
Quality Level
TopLine
|
Quality Level
StandardLine
|
Quality Level
StandardLine
|
Quality Level
TopLine
|
Quality Level
StandardLine, TopLine, TopLine Packages
|
| Closure Systems |
Closure Systems
Stopper + seal
|
Closure Systems
Stopper + seal
|
Closure Systems
Stopper + seal
|
Closure Systems
Stopper + seal
|
Closure Systems
Stopper + seal
|
Closure Systems
Stopper + seal
|
Closure Systems
Stopper + seal
|
Closure Systems
Stopper + seal
|
Closure Systems
Stopper + seal
|
Closure Systems
Stopper + seal or press-fit cap
|
Closure Systems
Stopper + seal
|
| Pre-washed, Pre-sterilized |
Pre-washed, Pre-sterilized
Non-sterile and sterile (adaptiQ®) available
|
Pre-washed, Pre-sterilized
Non-sterile and sterile (adaptiQ®) available
|
Pre-washed, Pre-sterilized
Non-sterile and sterile (adaptiQ®) available
|
Pre-washed, Pre-sterilized
Non-sterile available
|
Pre-washed, Pre-sterilized
Non-sterile available
|
Pre-washed, Pre-sterilized
Non-sterile and sterile (adaptiQ®) available
|
Pre-washed, Pre-sterilized
Non-sterile and sterile (adaptiQ®) available
|
Pre-washed, Pre-sterilized
Non-sterile and sterile* (adaptiQ®) available
|
Pre-washed, Pre-sterilized
Non-sterile and sterile* (adaptiQ®) available
|
Pre-washed, Pre-sterilized
Sterile packaging
|
Pre-washed, Pre-sterilized
Non-sterile and sterile (only TopLine in adaptiQ®) available
|
| Packaging | Packaging | Packaging | Packaging | Packaging | Packaging | Packaging | Packaging | Packaging |
Packaging
Bulk tray, adaptiQ® solution (tray, cup nest)
|
Packaging | Packaging |
| DATASHEET | |||||||||||
| Read More | Read More | Read More | Read More | Read More | Read More | Read More | Read More | Read More |
*available from Q2 2026
-
Vial coating process
-
Vial production process